|
|
Report of GHP Day Conference 1.12.2000 |
'Doing the right thing' - Prof. Nick Barber Need for value literacy However, there are conflicting priorities in the NHS structure. Utilitarianism is the greatest good for the greatest number. Autonomy is about individual choice. The Code of Ethics is necessary, but insufficient, as it does not go far enough. Core values for pharmacy There are four principles of health care ethics A suggested framework of values would have The priorities in healthcare are horizontal equity, where there is equal access / opportunity for everyone, and vertical equity, where it is possible to fast track people who are being discriminated against.
Prof. Barber spoke about the document which was available on the website of the Royal Pharmaceutical Society. It was intended to stimulate the debate on developing pharmacy values.
The point was made that safe, effective and economic treatments may not always be appropriate for an individual patient with individual needs. A value judgement is required. e.g. a non-formulary request for flurazepam for a terminally ill patient. However, values are hard to assess and express.
Looking at several sections of the report -
There are health policy dilemmas. The practice of pharmacy involves priority setting and policy decisions. We need to ensure that we embrace
This would be better entitled values at the core of pharmacy. Standards are provided, but are they any good? Often they are too vague to be useful. There are points for and against standards.

Development of judgement involves taking risks, assessing situations and learning from mistakes. Unfortunately, pharmacists are often too shy to develop this. They must accept uncertainty!
back to top  |
New Legislation on the Supply and Administration of Medicines - Ian Simpson The new legislation allows the sale, supply and/or administration of medicines, under certain conditions, by Sale, supply or administration must satisfy one or more of the following conditions The conditions apply as follows Patient Group Direction Patient Specific Direction Implications for the Supply Chain
The subject of this talk was the new legislation which was issued on 9th August 2000 after publication of the Crown Report. The report stated that the law should be clarified to ensure that health professionals who supply or administer medicines under approved group protocols are acting within the law. Dr June Crown herself said that if something is safe and sensible and benefits the patient, it should be legal.
B. In accordance with a Patient Group Direction
C. In the course of the business of the hospital
D. Pursuant to an arrangement made with an NHS body (i.e. Common Services Agency, Health Authority or Special Health Authority, NHS Trust, Primary Care Trust)
E. Individual supplying or administering must be a healthcare professional (i.e. nurses, midwives, health visitors, optometrists, pharmacists, chiropodists, radiographers, orthoptists, physiotherapists and ambulance paramedics)
This is a written direction relating to the supply and / or administration of a class of medicine to persons generally, signed by a doctor or dentist and by a pharmacist. There are detailed requirements for the content.
Here the medicine is supplied for the purpose of being administered to a particular person in accordance with the written directions of a doctor or dentist relating to that patient. The directions need not satisfy the detailed legal requirements for a prescription. Most hospital kardexes fall into this category.
The new legislation does have major implications for the supply chain, and all NHS bodies need to ensure that all supply and administration is legal as one of the statutes, HSC2000/026 carries the threat of legal action.
It was important to note that the new legislation clarifies the legal situation on supply and administration and is not about prescribing. It allows more flexibility and gives pharmacists a key role.
back to top  |
FWJ Hooper Award Oral Presentation 1
|
TTOs for Day Case / Short Stay Surgery Units - Improving the Service Caroline Elizabeth Elliott MRPharmS, Pharmacy Department, Selly Oak Hospital, Birmingham Caroline Elliott |
 |
Method:
An audit was carried out to investigate the prescribing of analgesics on D1 (day case unit) and C4 (short stay unit) for wisdom teeth removal and hernia repair operations using data obtained in the periods 1.1.99 to 10.2.99 and 1.5.99 to 30.6.99 respectively. A literature search was then undertaken:
Results:
The audit period showed variations in prescribing between clinicians and that the most appropriate analgesic was not always chosen. Using evidence based studies obtained from the literature search, guidelines and a protocol for prescribing analgesics were developed according to the type of surgery undergone and a new prescription form devised. These were agreed within a multidisciplinary setting involving consultants, practice development unit, pain management team and ward managers.
Discussion:
Different types of surgery were categorised according to the expected severity of pain (mild, moderate, and severe). Choices of analgesia were paracetamol for mild pain, co-codamol 30/500 for moderate pain and ibuprofen in combination with co-codamol 30/500 for severe pain. Pre-packs of these analgesics would be made available so that they can be issued from the ward by nursing staff.
Conclusion: Acknowledgments: References:
It is hoped that the objectives of this project will be met and a 3-month trial period is required to audit this project and assess the following:
I would like to thank Anne McGettigan, my project supervisor, for her help with this project.
1. Haddock J et al. Patient Controlled Oral Analgesia (PCOAH) for the management of post-discharge pain following day surgery. The Journal of One-Day Surgery, spring 1999: 3-8
2. Haynes TK, Evans DEN, Roberts D. Pain relief after day surgery: quality improvement by audit. The Journal of One-Day Surgery, summer 1995: 33-36
3. Kong KL, Donovan IA, Child DL, Nasmyth-Miller D. Demand on primary health care after day surgery. Ann R Coll Surg Engl 1997; 79: 291-295
4. Cooper SA. Five studies on ibuprofen for postsurgical dental pain. The American Journal of Medicine, July 1984: 70-76
5. Habib S et al. A study of the comparative efficacy of four common analgesics in the control of postsurgical dental pain. Oral Surg Oral Med Oral Path, November 1999; 70(5): 559-562
6. Ahlstrom U et al. The analgesic efficacy of diclofenac and ibuprofen in postoperative pain after dental extraction. Eur J of Clin Pharmacol, July 1993; 44(6): 587-588
7. Cooper SA et al. Ibuprofen and acetaminophen in the relief of acute pain: a randomised, double-blind, placebo-controlled study. J Clin Phamacol, Nov 1989; 29(11): 1026-30
8. Ahmad N et al. The efficacy of non-opiod analgesics for postoperative dental pain: a meta-analysis (abstract only). Anesth Prog 1997; 44(4): 119-26
9.Sagne S, Henrikson KF, Thilander H, Bertilson SO. Analgesic efficacy and side effect profile of paracetamol/codeine and paracetamol/dextropropoxyphene after surgical removal of a lower wisdom tooth. Journal of International Medical Research 1987; 15: 13-88
10.Kenneth C, Bentley DDS, Head TW. The additive analgesic efficacy of acetaminophen , 100mg, and codeine, 60mg, in dental pain. Clin Pharmacol Ther 1987; 42: 634-40
back to top  |
FWJ Hooper Award Oral Presentation 2
|
The Discharge Planning Pharmacist Krishna Patel*, Ann Jacklin** , Ofran Almossawi***, Katy Glynne****, *Medicine Directorate Pharmacist, **Chief Pharmacist, ***Discharge Pharmacist, ****Clinical Services Manager, Pharmacy Department, Hammersmith Hospital, London Ofran Almossawi |
 |
Introduction: Aim: Methods: Results: Discussion: Recommendations:
Hospital patients are often delayed at discharge due to a number of reasons. Waiting for discharge medication has often been mentioned as a reason of delay. In this project we set out to investigate whether having a discharge planning pharmacist was an effective method of speeding up the discharge of patients under the care of the medicine directorate. The service was operated as a pilot from December 1999 to March 2000.
The primary aim of this project was to speed up the discharge of patients under the care of the directorate of medicine. Secondary aims were to introduce a system of pharmacist’s transcribing discharge prescriptions, to counsel patients on the use of their drugs and to confirm the need to dispense continuing supplies of all medicines on discharge.
A C-grade pharmacist was appointed to the position of discharge pharmacist for a period of 6 months to set up this service.
The pharmacist then assisted with speeding up the discharge process by a variety of methods which included: transcribing discharge prescriptions, dispensing urgently required drugs, helping the dispensary to prioritise other patients and counselling patients which helped to identify drugs which did not need dispensing as patients had sufficient supplies at home.
Drug and Therapeutics Committee approval was gained to enable pharmacist transcription of TTAs. In addition, each participating consultant signed an "agreement to participate in the initiation of discharge medication by pharmacists" document.
Over the 4 month period of the pilot the discharge pharmacist was involved in 244 patients discharges (27% of all discharges). The pharmacist transcribed the TTA for 102 patients (42%) and for the remaining 142 patients (58%) the pharmacist was involved in counseling and scheduling.
On average the time from writing the TTA to patient discharge was 0.55 days for doctor written TTAs and 1.22 days for pharmacist written TTAs.
During the period of the pilot the discharge pharmacist recorded a total of 73 interventions relating to the 244 discharge prescriptions this represents an intervention rate of 1 query for every 3 prescriptions. 38 interventions (52%) arose directly from the novel service being provided by the discharge pharmacist and would not have been detected by the traditional services.
The results show that, in all aspects of the service monitored, there were many benefits to be accrued from the creation of a service providing a pharmacist dedicated to patient discharge. Benefits were shown for timelines of discharge prescribing, reduced dispensing costs and improved quality of prescribing.
It is estimated that the establishment of a discharge service with 1 dedicated pharmacist would be expected to speed and improve the quality of approximately 125 patients per month, which represent just over half of the patients discharged by the directorate of medicine each month and would be expected to reduced drug expenditure by up to Ł10,000 per annum.
It was recommended that the Trust should invest in creating discharge pharmacist positions within the Trust.
It was recommended that, as a minimum, 1 grade D pharmacist be appointed to Charing Cross and I grade D pharmacist be appointed to Hammersmith.
back to top  |
FWJ Hooper Award Oral Presentation 3
|
A comparison of two intravenous antibiotic reconstitution methods Presented by: Alison Eggleton MRPharmS, on behalf of Danielle Turner, Pharmacy Department Addenbrooke's Hospital, Cambridge Alison Eggleton |
 |
Objective: Design: Setting: Main outcome measures: Results: Conclusion:
To investigate the feasibility of replacing the current standard method of intravenous antibiotic reconstitution at Addenbrooke's Hospital, by comparing two novel reconstitution methods, the Baxter Minibag Plus (MBP) and the Macoflex Drug Reconstitution System (MTS).
A pre-trial telephone interview of nurses in other UK hospitals identified the MBP as a successful product. An 'on-ward' assessment was then conducted of ten cefotaxime and metronidazole reconstitutions for each novel method, at least one week after product introduction, and compared with results obtained for the current standard method. Assessment included time of preparation, interruptions and problems encountered. On conclusion of each four-week trial a questionnaire was completed by the individual users.
Two surgical wards at Addenbrooke's Hospital.
Reduction in reconstitution time with fewer errors and less interruptions, accompanied by an improvement in patient and staff safety.
Time savings of over 4.5 minutes per reconstitution were observed with the MBP and MTS. No problems were experienced with 20%, 40% and 70% of reconstitutions for the standard method, MBP and MTS, respectively. The average number of interruptions reduced from 2 ±
1.6 with the standard method to 0.2 ±
0.4 with the MBP and 0.3 ±
0.7 with the MTS. 61% of respondents for the MBP and 88% for the MTS found the product easier to use than the standard method. Over 82% of respondents agreed that the MTS offered the advantages of rapid preparation, easy preparation, no needle-stick injuries, minimal contamination and reduced handling. With the MBP, 61% of respondents experienced difficulty in connecting it and 83% experienced air in the infusion line, compared with 12% of each respectively for the MTS. 61% (MBP) and 94% (MTS) favoured introduction of that product to Addenbrooke's Hospital. The cost saving per dose of the MBP was 17p and of the MTS 96p.
It is recommended to introduce the Macoflex Drug Reconstitution System (MTS) to Addenbrooke's Hospital, as an intravenous antibiotic reconstitution method, in order to improve patient and staff safety. Additional benefits would be significant time and cost savings, a decrease in sharps material generated, retention of the possibility of multiple dose administration and high acceptability by staff.
back to top  |
FWJ Hooper Award Oral Presentation 4
|
A review of TPN prescribing at North Tyneside General Hospital Philippa J Burch & Steve Williamson Pre-registration Pharmacist & Senior Pharmacist - Aseptic and Cancer Services Pharmacy Department, North Tyneside General Hospital, Tyne and Wear, NE29 8NH Philippa Burch |
 |
The current TPN service provides a range of standard bags, which can be amended to suit individual patients. Patients are identified by clinicians and although there is no standard pathway of care most refer their patients to a dietician for assessment prior to prescribing. Pharmacy aseptically prepares the TPN, monitors the patients’ electrolytes, checks stability and advises prescribers on regimen changes when appropriate. It was decided to review the use of TPN in the hospital to identify current practice with the aim of improving the service. Objectives: Method:
Patients receiving TPN during a twelve month period were identified from departmental records. Patient information collected included patient demographics, indication for feeding, number of treatment days, nutritional assessment and monitoring performed. Data was compared to ASPEN guidelines1.
Results:
| Number of patients receiving TPN | 48 |
| % of patients receiving nutritional assessment | 84 |
| % of patients receiving TPN for < 7 days | 55 |
| % of patients receiving TPN for < 5 days | 33 |
| % of patients receiving full baseline tests prior to treatment | 8 |
| % of patients receiving daily U&Es | 35 |
| % of patients receiving twice weekly monitoring (LFTs, FBC, calcium, phosphate) | 76 |
Discussion: The indications for TPN were all correct due to their inclusion in the ASPEN list, however as this was a retrospective audit there was not the ability to assess the clinical judgement as to the severity of the condition. For example cancer is included in the list but only in extreme cases where malnourishment is proven, Cancer patients in this study had no documentation as to their nutritional need for TPN. It was shown that patients were routinely being treated for less than the minimum 5 days and the recommended 7 days. This is inappropriate as the risks of centrally administered TPN outweigh the benefits if patients are not treated for at least 5 days. A small number of patients did not receive TPN for the recommended minimum duration due to death or transfer to other hospitals. This would slightly bias the results but despite this substantial numbers were not treated for an appropriate length of time. Patient monitoring did not meet the recommended standards. 92% of patients did not receive a full baseline, 65% did not receive daily U&Es and 24% did not receive twice weekly tests. It is essential that patients receive correct monitoring to prevent metabolic disturbances, excess nitrogen administration and fluid overload. Conclusion: References:
Malnutrition in hospitalised patients has been shown to be as high as 65% and these patients are more likely to suffer complications, more susceptible to infection and experience delayed wound healing2. TPN can have a major impact on patient care when prescribed appropriately but if not used correctly can result in an increased incidence of TPN related complications. The most common of these are catheter infections, thrombophlebitis and pnuemothorax resulting from incorrect administration. Metabolic imbalances and electrolyte disturbances can result from incorrect prescribing of TPN and inadequate patient monitoring.
16% of patients did not receive a nutritional assessment and it is possible that these patients did not require TPN or were prescribed the wrong formulation.
The areas highlighted by this study show that patients prescribed TPN are not receiving the level of care required. This supports the need for the introduction of a multi-disciplinary nutrition team, as all patients would then receive a full assessment, recommended baseline tests, daily reviews and all necessary monitoring.
1. American Society for Parenteral and Enteral Nutrition (ASPEN) Guidelines for the use of Parenteral and Enteral Nutrition in Adult and Pediatric Patients. J Parent Ent Nutr 1993; 17(4 Suppl)
2.Murphy A, Scott A. Artificial nutritional support - what are the options? Hosp Pharm 2000; 7:146-154
back to top  |
Reducing Risk - Peter Pope, Manager Human Factors, Britannia Airways What can pharmacy learn from the airline industry, and what can aviation learn from us? Apparently, shift handovers in hospitals are supposed to be the best, and that is what airlines aim for, so we can learn from each other.Medicine really began with Hippocrates in 460BC. When did aviation start? The first aviation accident was probably Icarus flying too close to the sun. What would the Air Accident Investigation Unit have said there? Was it a design problem? Was it an operational or procedure problem i.e. should the pre-flight warning about flying too close to the sun have been more forcefully given? Or was it a maintenance error. |
 |
The major problem to be overcome by an aeroplane is gravity. Air Traffic Control (ATC) helps to avoid problems in the air. Cabin crew avoid problems with the passengers. Maintenance keep the plane well serviced and fix any problems. Refuelers ensure the correct quantity and type of fuel is used. Baggage handlers ensure that luggage is loaded in the right places and in such a way as to avoid any part of the aircraft being overloaded. The aviation industry works within a tight legislative framework. The1944 Chicago Convention allows airlines to fly into other countries without fear of being shot down. The International Civil Aviation Organisation (ICAO) sets standards and recommends practices. The Civil Aviation Authority (CAA) ensures compliance in the UK. The EU Joint Aviation Authority is trying to harmonise the regulations across Europe. Human factor training requirements feature in several pieces of legislation, but the aviation industry cannot comply with them since it doesn't know what the human factors are! To prove that regulations are merely a record of previous accidents and incidents, details were given of three accidents and the regulations introduced after each. Human error is the causal factor in 80% of aviation accidents and incidents, and the humans involved are designers, manufacturers, flight crews, cabin crews, ATC controllers, dispatchers, ground handlers and maintenance staff. Designers are still creating aeroplanes where it is still possible to cross connect systems. If a component is damaged during the manufacturing process, it is often repaired and then fitted. British Airways have a QA department at Boeing in Seattle to ensure no repaired components are fitted to their new aircraft. Flight crews cause the majority of accidents and incidents. Cabin crew either don't see problems outside the aircraft or don't say. Ground handling staff such as refuelers and caterers can cause problems e.g. by ramming aircraft with their vehicles or anything their vehicle is towing. Maintenance staff can accidentally cross connect systems. Various safety enhancements have been introduced such as new zero flight time simulators which are so advanced that a pilot should be able to go straight from there to the cockpit without any trial flights. Britannia uses these but also include trial flights in the training. Engine stall warning devices, traffic collision avoidance systems and enhanced ground proximity warning systems are others now in use. The current focus within the industry is on the following areas Looking at the causes of errors over the years, we see It is important that everyone is aware that accidents and incidents caused by human error do not respect status, length of service or experience of the individual. So what will a human factors programme achieve? It should lead to safety and performance improvements; reduce the risk of becoming a statistic; provide 'corporate comfort'; demonstrate 'due diligence'; reduce the possibility of litigation - written evidence of discussing such issues is a good defence; increased confidence of staff and public and helps to prepare for future legislation. What are the contributing factors for errors? Management errors are mostly latent errors such as policy or procedural mistakes. These may lie dormant for months. They make few active errors. The problem is that latent and active errors combine to make an accident or incident. Professional cultures have to be taken into consideration. Comparing pilots and maintenance engineers, Pilots have a strong motivation to succeed and pride in their profession. Unfortunately, they also have a sense of personal invulnerability, and are sure that they don't make errors under high stress. They see themselves as good decision makers in emergencies, not reliant on previous experience, and don't think that personal problems affect their performance. Engineers are dependable, modest, self sufficient and willing. They are proud of their integrity, distrust words - preferring actions and dislike sharing thoughts or asking for help. They prefer to think for themselves and tend to be loners. For all these reasons they very frequently put themselves in risky situations. For every major accident, there are 40 serious incidents (e.g. diverted aircraft, damage to aircraft etc.) and 600 other events. It is impossible to deal with everything, so it is important to prioritise. It is often a number of other events which contribute to a serious incident. Therefore a full investigation of a serious incident and rectification of the causes may resolve many of the other events without effort. Once all the serious incidents are being fully investigated, it is possible to move on to the lower levels. What are the essentials for any HF programme? There must be management buy-in. Without it, nothing will happen. A non-punitive disciplinary policy must be in operation. Britannia Airways estimate that less than 5% of their disciplinary cases result in punitive action. There must be an open error reporting process. As soon as the process becomes confidential, it signifies that there is something to fear. There should be a systematic incident review process including contributing factor analysis. There should also be corrective action implementation, audit process and written procedures. In essence - it is a full time job! The next steps are Recommendations for success start with the appointment of a credible HF facilitator who will establish why it is important and how to implement. There is a need to gain management confidence and then obtain management commitment. The training needs to be relevant to the organisation and staff cultures. Then comes the development of HF processes and procedures, which are important to ensure that in the event of the HF manager leaving, all the previous work is not wasted. It is important to communicate successes. And remember, it is a cultural evolution, not revolution. The main obstacles are wavering management commitment, lack of resources, convincing the sceptics and dinosaurs, maintaining momentum and dealing with maverick managers. In summary, all humans are error prone and several factors combine to cause errors. Usually these factors are organisational and can be managed. Management commitment is key and doing nothing is not an option.
back to top  |
Closing Comments and winners presentations - Helen Remington, President, GHP
The President closed the Day Conference by presenting the FWJ Hooper Award to Alison Eggleton, and the poster award to Nicholas Duncan for his poster entitled 'An audit of laxative prescribing in patients receiving opioid analgesics'.

FWJ Hooper Award presentation |

Poster Award presentation |
back to top  |
back to top  |
Poster 1
Discordance: Coronary Care Patients’ Knowledge of Medicines
Ann-Dora N Mundia, Anthony R Cox & Dr John F Marriott *
Pharmacy Department, City Hospital NHS Trust, Dudley Road, Birmingham, B18 7QH
*Senior Lecturer, Pharmacy Practice Group, Aston University, Birmingham
Introduction: Method: Results:
The National Service Framework (NSF) for Coronary Heart Disease (CHD) sets out clear objectives for the prescribing of medication to patients with CHD1. Concordance aims to help patients and prescribers to make informed choices about diagnosis and treatment and optimise the potential benefits of medical care2. Knowledge of medication has been shown to help reduce changes in medication regimes3. This study aimed to investigate coronary care unit (CCU) patients’ knowledge about their medication and the manner in which they were informed of the commencement of new medication.
Semi-structured interviews with CCU patients prior to discharge. Questions were asked about the name, purpose, and timing of their medication and compared to the charted medication. Patients were asked if they were aware of new medication and how they were informed of the change. Specific questions regarding the use of sublingual glyceryl trinitrate (GTN) preparations were asked.
50 patients were interviewed (37 male and 13 female) with an average age of 58 (standard deviation 14). Main diagnoses included: unstable angina (20), myocardial infarction (15), atrial fibrillation (8), and heart failure (4). Prior to discharge the average number of medicines prescribed to each patient was 6 and the average number of new medicines commenced per patient was 2.
 |
Patients’ knowledge of their medication was highly variable. (Figure 1). 40 patients were commenced on new medication. 24 were informed of the change by hospital staff, 3 were unaware of the change and 13 discovered the change themselves upon being administered their medication. 38 patients were using sublingual GTN preparations. Many were unsure of the course of action to take if sublingual GTN had no effect, with only 27% stating they would ring for an ambulance. Discussion: References:
Our study shows that there is a large deficit in knowledge about medication regimes in the patients surveyed. Many patients have new medication prescribed prior to discharge and a significant number are not being informed about these changes which could impact on their willingness to take the prescribed medication regime . Lack of knowledge of the correct course of action to take in the event of failure of GTN preparations is particularly worrying. Increased nurse and pharmacist involvement in patient education and a self-medication scheme may facilitate improved knowledge and allow patients to take ownership of their medication regime4.
(1) Department of Health. National Service Framework for Coronary Heart Disease. London, 2000.
(2) http://www.concordance.org (accessed on 16th October 2000)
(3) O'Neill CK, Poirer TI. Impact of patient knowledge, patient-pharmacist relationship, and drug perceptions on adverse drug therapy outcomes. Pharmacotherapy. 1998;18(2):333-40.
(4)Noy K. A self-medication scheme in a post-coronary care unit. British Journal of Nursing. 1997;6(7):369-374
Acknowledgements: We would like to thank Mr Chris Anton, administrative co-ordinator for the West Midlands Centre for Adverse Reaction Monitoring for his assistance with this project.
back to top  |
Poster 2
The role of the community services pharmacist in the implementation of nurse prescribing Redbridge and Waltham Forest experience
Ms Kalpana Patel & Mrs Kim Churchman
King George Hospital Goodmayes
Introduction / Background: Local Implementation: Guidelines: Training: Support Groups: Joint Formulary: The Way Forward: References:
Following the recommendations of the Cumbeledge report (1986) the roll out of Nurse Prescribing was announced by the Secretary of State on 20th April 1998.
Detailed guidance was then issued in December 1998 (HSC 1998/232). Nurse Prescribing has been widely advocated as a way of providing a more flexible service that is responsive to patients needs. The implementation of Nurse prescribing is based on the recommendations of the Crown report (Department of Health, 1989) which identifies a number of potential benefits in the significant improvement in patient care. Nurse Prescribing will enable nurses to adopt a more holistic approach to patient care and promote increased collaboration and cohesiveness within Primary Care.
Nurses working in the community who hold Health Visitor or District Nurse qualifications and upon completion of the approved training course may prescribe from the items listed in the Nurse Prescribers’ Formulary. This process was managed by multi-disciplinary Steering Group led by the Health Authority and local co-ordinating group which comprised of Nurses, (Health Visitor and District Nurse ) Pharmacist and a Project Administrator.
The procedure workshop group developed guidelines to set standards for good practice and have uniformity across the Health Authority. The Prescription form, Accountability, Reporting of Adverse drug reactions, Security and Safe Handling of Prescription forms and record keeping were some of the many topics addressed.
Although the Individual University supplied the formal training, contingency plans were in place to offer any extra support should the need arise, this could be accessed via the lead nurse.
Update sessions were offered to qualified prescribers to boost confidence and share any individual concerns they may have had before actually commencing prescribing.
These were initiated by the steering groups to share practice and discuss any problems. The Pharmacist would relay information and provide continuing education on related topics.
A multi-disciplinary group developed a local formulary. This is to be used throughout the Health Authority and will be reviewed regularly. Specialist advice was sought from various disciplines before deciding on the product range. This group will address entry of new products and any change in practice.
This can be summarised as follows:
1. Department of Health - The Cumberledge Report London HMSO 1986
2. Department of Health - Crown Report 1999
3. Nurse Prescribing - A guide for Implementation Department of Health Dec 98
4. Guideliens for records and record keeping - UKCC 1998
5. Nurse Prescribing Procedures Thameside Community Health Care Trust
6. Nurse Prescribing Humphries and Green 1999
back to top  |
Poster 3
Drug use evaluation of tinzaparin in patients with suspected DVT - multidisciplinary approach
Rosaline Meju BSc Pharmacy DipClinPharm MRPharmS
Clinical Effectiveness Pharmacist. Pharmacy Department Leicester General Hospital, Leicester LE5 4PW
Background: Objectives: Methods: Results: Discussion and Achievements: Conclusions: Acknowledgements:
After presentation of the above information to the Medicines Management Group it was agreed to carry out an audit with the following objectives:
2. Delay in Venography
3. Tinzaparin not crossed off when therapeutic INR is reached
Results were presented at a medical audit meeting where it was agreed that:
Members of Medicines Management Group: Dr. Tom Robinson, Dr.S.Evans, Dr. S.Dawson, Mrs l. Webb, Senior Nurse, Jenny Powell, SSN Cath Oxley, Senior Pharmacist, Ros Meju, Clinical Effectiveness Pharmacist. Claire Honeybourne, Clinical Librarian
Louise Garbett, Ward Manager. Graeme Hall Principal Pharmacist Clinical Services
back to top  |
Poster 4
An investigation of cisplatin prescribing in patients with renal impairment
Nicholas Duncan,
Pharmacy Department, Queen Elizabeth Hospital, Birmingham
This study aimed to investigate the prescribing of cisplatin in patients with renal impairment and to determine the effect of the drug on renal function. Pharmacy records were used to obtain data for all patients who started a cisplatin-containing chemotherapy regime between July and December 1999. For each patient, details of cisplatin dose and renal function (calculated using the Cockcroft and Gault equation) for each course of chemotherapy were recorded. Data were collected for 171 patients, who received a total of 582 courses. The most commonly prescribed regime was MIC (Mitomycin C, Ifosfamide and Cisplatin) which was given to 75 patients (240 courses). 47 out of 163 evaluable patients had a calculated GFR of <60ml/min prior to their first course of cisplatin. Eight of these (17%) received a dose reduction of cisplatin. For all courses of cisplatin, 195 out of 542 patients had a GFR of <60ml/min, of whom 61 (31%) received a dose reduction. The average reduction was 25%. 22 patients received cisplatin despite a GFR of <40ml/min, with 6 of these patients receiving no dose reduction. Patients with renal impairment who received >1 course of cisplatin were evaluated to determine the effect of the drug on subsequent renal function. In those patients who received a dose reduction, GFR decreased by an average of 1% (95% CI: 1.9% increase - 3.9%decrease) between each course. In those patients who received no dose reduction, there was, on average, a 4% decrease in GFR (95% CI 1.5%-6.5%). Similar trends emerged for the sub-group of patients who received MIC chemotherapy. It was concluded that the majority of patients with renal impairment were not receiving a dose reduction of cisplatin. Such patients tended to have a greater decrease in renal function after each course when compared with those who did receive a dose reduction, although the trend did not reach statistical significance.
back to top  |
Poster 5
An audit of laxative prescribing in patients receiving opioid analgesics
Nicholas Duncan, Sarah Jones and Susan Whitmarsh.
Pharmacy Department, Queen Elizabeth Hospital, Birmingham
Local guidelines recommend that prophylactic laxatives should be prescribed for all patients who are started on a weak or strong opioid, with senna or co-danthramer being the agents of choice. This study aimed to determine firstly what proportion of patients receiving a regular opioid were prescribed a laxative and secondly what laxatives were being used. Over a four week period, data were collected on laxative prescribing for all oncology in-patients receiving a regular opioid. Data were collected for 50 patients, 44 (88%) of whom were receiving a strong opioid. Laxatives were prescribed for 29 (58%) of these patients. Of the 21 patients who were not receiving a laxative, documented reasons for this omission could be found for 6 patients. Co-danthramer was the most commonly prescribed laxative (22/29 patients), closely followed by lactulose (17/29 patients). Only 4 patients were prescribed senna while no patients received co-danthramer strong. 14 patients (48%) were receiving a combination of co-danthramer and lactulose. It was concluded that a significant proportion of patients receiving a regular opioid were not being prescribed an accompanying laxative. Lactulose appeared to be over-prescribed, especially in combination with low doses of co-danthramer. It was also concluded that there was scope for increased prescribing of senna and co-danthramer strong. Following this audit, new guidelines for laxatives prescribing in patients receiving opioid analgesics were introduced by the oncology directorate.
This Poster won the Poster Awardback to top  |
Poster 6
Validation of cleaning methods and effects on environmental monitoring
Julian Smith,
Pharmacy Quality Control, Burton Hospitals NHS Trust.
Microbiological environmental monitoring of hospital pharmacy clean rooms was undertaken. Results following cleaning with a hospital detergent and single bucket system were compared to results with a triple bucket system and commercial clean room disinfectants. The classification of clean rooms investigated included, a non-sterile production room (BS 5295 part 1 classification K), an aseptic isolator dispensing room (BS 5295 part 1 classification K) and a sterile production room (BS 5295 part 1 classification H). The microbiological environmental monitoring included, contact plates, settle plates, active air sampling and viable microbiological counts of cleaning solutions. In the non-sterile and aseptic dispensing rooms, a triple bucket with hospital detergents made slight improvements in the environmental results over a single bucket. These improvements were not statistically significant. The use of both a triple bucket and Shield Medicare disinfection solutions statistically improved the environmental results in the non-sterile (P < 0.01) and aseptic dispensing (P < 0.05) rooms. This system did not significantly change the environmental results in the sterile production area. The lack of improvement was probably due to the high standard of results obtained using the original cleaning method.
In summary the use of a triple bucket and commercial clean room disinfectants significantly improved the environmental monitoring results over the traditional cleaning methods used in the non-sterile and aseptic isolator preparation rooms. A significant change was not seen in the sterile preparation room, because of the low numbers of viable counts obtained with all cleaning methods.
back to top  |
Poster 7
The development of a competency-based training programme for dispensers at the Royal Liverpool University Hospital (RLUH).
M.T. O’Hare*, A.J. Golding**, J.A. White ***, R. Horner ****
Pharmacy Department, Royal Liverpool & Broadgreen University Hospitals NHS Trust, Liverpool
*Dispensary Manager, **Principal Pharmacist - Medicines Management, ***Chief Technician, ****Director of Pharmacy
Introduction: Aims: The training and certification programme: Results: Conclusion:
Staffing pressures and financial restrictions in the NHS have highlighted the need to review the appropriate deployment of staff to various duties. As pharmacists have moved into new roles, pharmacy technicians have themselves adopted duties once performed by pharmacists, including audit, purchasing, drug information and the final checking of prescriptions. At the RLUH, it was believed that there was a role for suitably trained and certified staff without a Nationally recognised qualification to adopt roles in dispensing. A competency-based training and accreditation scheme was developed and introduced.
The aim of the project was to develop and pilot a training and accreditation programme for unqualified staff to perform safe and accurate dispensing. As a result, it was intended to improve job satisfaction and career progression for these staff, and release pharmacy technicians for new roles, notably final dispensing checking and patient counselling.
The programme adopts the competency based model with evidence collection familiar to pre-registration pharmacists and student pharmacy technicians. At the start of the programme, trainees are introduced to the concept of evidence collection. Four dispensing units are undertaken, each including several elements. The trainees undertake these units in the following order: in-patient dispensing, discharge prescriptions, out-patient prescriptions and finally, basic extemporaneous prescriptions. Each unit includes dispensing activities as well as related tasks such as ordering stock, generating labels etc. In parallel with these four units, a common skills unit is completed which includes time management, communication skills, use of information sources and similar activities. Supervision of evidence collection is the responsibility of the senior technicians, and assessment and accreditation is carried out by the Technician Dispensary Manager. A Certificate signed by the Chief Pharmacist is presented upon satisfactory completion of the programme. The whole programme can be completed in about six months, although trainees with community pharmacy experience will proceed much faster.
In the two years since the programme was introduced five trainees have been recruited; most of these have had some prior community pharmacy experience. Three trainees have satisfactorily completed the programme, and of these one has progressed to student pharmacy technician training and the other two are currently working as dispensers whilst studying for GCSE qualifications to do the same. Unfortunately, the other candidates did not complete the course, but found their partial training in hospital a useful springboard to the National Pharmaceutical Association training programme with a well-known multiple community pharmacy chain.
The RLUH has successfully implemented a training programme which gives opportunities for career progression to unqualified staff. By implementing this programme the RLUH has empowered and added career satisfaction not just to the dispensers, but the pharmacy technicians who have adopted checking roles, as well as a wide variety of other functions. This in turn has released qualified pharmacists to concentrate on their clinical roles and professional development rather than dispensary activities, which should improve recruitment and retention in a difficult national situation.
back to top  |
Poster 8
Adherence to the George Eliot Hospital NHS Trust 'anti Clostridium difficile' respiratory antibiotic guidelines
Lee Stewart BSc PhD MRPharmS, Clinical Pharmacist
Pharmacy Department, George Eliot Hospital, Nuneaton
Introduction:
Antibiotic-induced Clostridium difficile infection causes considerable patient suffering and mortality.1-3 The diagnosis, treatment, support and nursing of sufferers and the strict environmental control procedures required during an outbreak represents a significant drain on hospital resources.3-5
In 1998 an audit was conducted to examine C. difficile infection within the George Eliot Hospital. This audit linked C. difficile infection with the use of broad spectrum antibiotics (particularly cefuroxime and co-amoxiclav) in the treatment of lower respiratory tract infections (LRTIs). Subsequently, these antibiotics were removed from the hospital's formulary guidelines for the treatment of LRTIs; the guidelines were revised with the aim of treating LRTIs effectively whilst minimising the risk of C. difficile infection. Pharmacists and prescribers were informed of the changes and the results of the audit via a news letter.
More recently, anecdotal reports from clinical pharmacists suggested that the revised guidelines were not being followed and that the use of cefuroxime and co-amoxiclav in the treatment of LRTIs was increasing. Consequently, a new audit was conducted to examine the extent to which the guidelines were followed.
Method:
The audit was conducted retrospectively on patients admitted during the period of December 1999-January 2000. Data were obtained from the medical records of 25 patients admitted with exacerbations of chronic obstructive pulmonary disease (COPD) and 26 patients admitted with pneumonia. Patients were excluded if their medical records were not available or incomplete. All patients included were 18 years old or older (since the formulary guidelines are not applicable to paediatric patients). The following data were collected for each patient:
Results:
The key findings were:
Conclusion:
These results fell short of the standards set for the audit and it was concluded that the formulary guidelines for the treatment of LRTIs should be adhered to more frequently. Possible explanations for the poor adherence include prescribers being unaware of the guidelines or being unwilling to follow them. A re-education programme (aimed at doctors and pharmacists) is under development to raise the profile of the hospital guidelines and the rationale behind them. Prescribing in this area will be re-audited once the re-education programme has been completed to ensure that LRTIs are treated effectively whilst minimising the risk of C. difficile infection.
References:
1. MacLaren R, Morton TH, Kuhl DA. Effective management of Clostridium Difficile colitis. Hosp Pharm 1997; 32: 1126-32.
2. Buckley P. The treatment of CDIC. Pharmacy in Practice 1996; 48-9.
3. Spencer RC. Clinical impact and associated costs of Clostridium Difficile-associated disease. J Antimicrob Chemother 1998; 41 (Supp C): 5-12.
4. Wilcox MH. Antibiotics and Clostridium Difficile infection. West Sussex: Eurocommunica Publications.
5. Wilcox MH, Cunniffe JG, Trundle C, Redpath C. Financial burden of hospital-acquired Clostridium Difficile infection. J Hosp Infection 1996; 34: 23-30.
back to top  |
Poster 9
The Role of the Directorate Liaison Technician
Edwards L, Bamford S, Thomson S
Pharmacy Department, North Staffordshire Hospital NHS Trust.
Introduction: Objectives of post: Results: Conclusion:
The North Staffordshire Hospital Trust (NSHT) is a 1,300-bed hospital providing a range of both acute and specialist services. In 1997-98 the Trust’s drug budget of Ł5.7 million was more than Ł800,000 overspent. This prompted a review of the systems in place to control drug expenditure. As a result of this a comprehensive Medicines Management system was implemented. This involved: establishment of Medicines Management Group (MMG); and dedicated pharmacist post to review high cost drugs (> Ł15,000 per annum); and a named pharmacist attached to each directorate. All directorate pharmacists liaise on a regular basis with the business manager, clinical director and accountant within their directorate. This work lead to a rapid increase in the number of requests for detailed drug expenditure and usage data. This was seen as an opportunity to develop an extended role for the pharmacy technician, and in May 1998 a full-time MT03 Directorate Liaison Technician (DLT) was appointed.
The results show improved production, interpretation and dissemination of drug usage and expenditure information to pharmacists and directorates since the introduction of the directorate liaison technician role. The development of a comprehensive medicines management system has resulted in the drug budget going into an underspend situation for the last 2 years (Ł21,000 underspent on a budget of Ł6 million in 1998/99 and Ł40,000 on a budget of Ł6.4 million in 1999/00). The post is viewed as a vital component of this system. As the service has developed demands for directorate support and information have increased. The Management Board has acknowledged this, and in April 2000 a business case for a second Directorate Liaison Technician with full time secretarial support was approved.
back to top  |
Poster 10
The stability of diamorphine and glycopyrrolate in PCA syringes
Smith J*, Hirsch C, Marriott J, Wilson K and Nash S
Pharmacy Practice Group, School of Pharmacy, Aston University, Birmingham B4 7ET. *Pharmacy Department, City Hospital, Birmingham B18 7QH
Objectives Methods Results Conclusions Reference
Diamorphine and glycopyrrolate mixtures are frequently administered by subcutaneous infusion to patients with terminal disease in order to alleviate both pain and excessive secretions 1. However, little is known about the chemical stability of these agents when mixed. The present study was designed to examine the chemical stability of glycopyrrolate and diamorphine mixtures contained in patient controlled analgesia (PCA) syringes.
HPLC assays were based upon the United States Pharmacopoeia (24) method for glycopyrrolate injection and were performed using a Hewlett-Packard HP1100 instrument with auto sampler, a variable wavelength detector (G1314A) and a 4.6x250mm licrosorb RP Select B column. Detection wavelength was set at 232nm and column oven temperature at 40oC. The compositions of test solutions were selected to mimic the dose forms currently administered in clinical practice over a 24-hour period1 (0.6mg glycopyrrolate and >15mg diamorphine). Three test solutions were prepared; all contained 0.6mg glycopyrrolate in 8ml combined with 5mg, 90mg or 200mg of diamorphine in a plastic syringe (Beckson and Dickson). Syringes were sealed with a blind hub (Braun) and stored in the dark at 32oC prior to assay with 3 repeats at 0, 2 and 7 days. Assays were found to be linear (diamorphine r=0.9998, glycopyrrolate r=0.9994) and reproducible within day (diamorphine %RSD=1.62, glycopyrrolate %RSD=1.91) and day-to-day (diamorphine %RSD=0.44, glycopyrrolate %RSD=2.47).
In the mixture containing 200mg diamorphine, concentrations of glycopyrrolate were reduced by 2.4% (to 0.654 ± 0.005mgml-1) and 6.0% (to 0.630 ± 0.003mgml-1) at 2 and 7 days respectively whereas, diamorphine concentrations were reduced by 3.5% (to 187.3 ± 1.0mgml-1) and 9.3% (to 176.1 ± 0.6mgml-1) respectively at these times. In the mixture containing 90mg diamorphine, concentrations of glycopyrrolate were reduced by 1.0% (to 0.605 ± 0.002mgml-1) and 0.7% (to 0.615 ± 0.002mgml-1) at 2 and 7 days respectively whereas, diamorphine concentrations were reduced by 3.5% (to 86.3 ± 0.7mgml-1) and 10.7% (to 79.9 ± 0.6mgml-1) respectively at these times. In the mixture containing 5mg diamorphine, concentrations of glycopyrrolate were reduced by 4.4% (to 0.591 ± 0.001mgml-1) and 0.5% (to 0.615 ± 0.006mgml-1) at 2 and 7 days respectively whereas, diamorphine concentrations were reduced by 12.9% (to 4.19 ± 0.02mgml-1) and 15.2% (to 4.08 ± 1.08mgml-1) respectively at these times. No significant consistent changes in sample pH were observed over the storage period.
The present study indicates that at the concentrations currently used in clinical practice, mixtures of diamorphine and glycopyrrolate would be chemically stable in PCA syringes for 2 days according to a nominal 10% end of shelf life limit. Samples were stored at 32oC to simulate conditions of use rather than refrigerated storage. Our study also showed that after 7 days, there was some evidence of degradation of diamorphine. Further studies are necessary to define the total viable storage periods for these solutions but our results suggest that freshly prepared solutions currently used in clinical practice would undergo minimal chemical degradation during the normal 24-hour administration period.
1. British National Formulary (no 38) London British Medical Association and RPSGB 1999
back to top 
Poster 11
Quinolone resistance patterns in a large district general hospital
Curtis C*, Marriott JF and Wilson KA
Pharmacy Practice Group, School of Pharmacy, Aston University, Birmingham B4 7ET. *Pharmacy Department, Queens Hospital, Burton on Trent DE13 0RB.
Introduction Method Results Discussion Reference
Antibiotic resistance is recognised as a major problem in healthcare1. Recent recommendations to curb increasing resistance patterns include the use of strategic systems for resistance surveillance1. However, in many secondary healthcare sites, resource and technological barriers compromise the achievement of this aim. The present study describes the use of data from a national pilot of an electronic patient record system in a large district general to assess patterns of quinolone antibiotic use and resistance.
A systematic drug use review was conducted for quinolone antibiotic administered between January and October 1999 inclusive. Data was generated by a hospital integrated information system (Medical Information Technology Inc. MA, USA), which facilitates the correlation of patient microbiological testing procedures with prescribing.
During the study period, 879 patients were prescribed a quinolone antibiotic. Almost one third (29%) of these cases were in the Elderly Care speciality. Together with General Surgery, Out Patients and General Medicine these specialities accounted for 633 (72%) of patients treated with a quinolone. Microbiological sensitivity testing was undertaken in only 568 (65%) of the cases. Organisms were isolated from only 311 (55%) of the samples and of these 283 (91%) were sensitive to a quinolone. However, 12 isolates (4%) were of intermediate resistance and 16 (5%) were quinolone resistant (MIC > 2m
gml-1). Nearly half (44%) of the resistant organisms belonged to Pseudomonas spp. The resistant or partially resistant isolates arose from patients in Elderly Care (43%), General Medical (29%), General Surgical (18%) and Orthopaedic (11%) locations.
Recent national advice has indicated that antibiotic use and resistance patterns should be audited regularly and that prescribers should be provided with appropriate education1. Currently, time constraints and limitations of information technology systems render such aspirations impractical. However, the patient information system utilised in the present study facilitated rapid and complete data gathering. The results indicate that, despite advice to the contrary, quinolones are often prescribed "blind". Moreover, resistance to quinolones is encountered, albeit at present only in a relatively small number of patients. If this behaviour is duplicated in other hospitals then clinical pharmacists should ensure that they check whether sensitivity testing has been carried out when they review prescriptions for quinolone antibiotics. These problems were most prevalent in Elderly Care, possibly because of longer than average stays during which serious infection may be acquired. It is suggested that targeting antibiotic prescribing in the Elderly might therefore result in the greatest impact upon drug expenditure and increased resistance patterns.
1. Standing Medical Advisory Committee (Department of Health). The path of least resistance. Main report. London: Department of Health, 1998
back to top 
Poster 12
Quantitative analysis of minoxidil lotion by FT-IR
Raynor D and Smith JC
Pharmacy Department, Queens Hospital, Burton on Trent DE13 0RB.
Introduction Method FT-IR determinations: spectra were recorded using a Nicolet 460 Protégé FT-IR equipped with a DTGS KBr detector, KBr beamsplitter, spectratech ARK smart accessory with zinc selenide "trough" crystal having an incidence angle of 450 providing 12 multiple reflections. All spectra were taken in ambient laboratory conditions using, 32 co-added interferograms, backgrounds taken directly before each spectra, Happ-Genzel apodization, a resolution of 4cm-1 giving a collection time of 39 seconds, a spectral range of 4000 - 650 cm-1 and no additional spectral smoothing. Preparation of standards: prepared using Class A glassware to measure volumes and a four figure analytical balance used for weighing. The strengths of minoxidil in the standards ranged from 2%w/v to 5%w/v. A blank was prepared containing all ingredients except minoxidil powder. Preparation of samples: samples of minoxidil lotion used were from previously manufactured batches and sampled without further preparation. Analytical determinations: each strength of lotion was assayed in triplicate, to assess reproducibility. The standards were applied to the zinc selenide trough in turn, after each strength had been scanned the trough was cleaned with IMS 99%, wiped and allowed to dry. This procedure was repeated for all four standards and the blank lotion. All spectra, including standards and lotion, had the blank spectra subtracted. The subtracted spectra were saved and imported into and analytical software package called "Tq analyst" (version 1.2). This software automatically uses given parameters to create a calibration curve and method. The given parameters were, simple beers law as the analysis type, constant path length, minoxidil as the named component, units of %w/v and a non zero intercept. The calibration standards consisted of subtracted spectra of minoxidil standards with their actual concentrations. Results and Discussion References 2. Reynolds JEF Editor Martindale, The Extra Pharmacopoeia 32nd edition. The Pharmaceutical Press 1999.
Infra-red (IR) spectroscopy has traditionally been associated with the identification of pharmaceutical raw materials and finished products. With the advent of Fourier-Transform Infrared (FT-IR) spectroscopy and modern software IR spectroscopy is increasingly used as a quantitative method 1. The object of this study was to investigate the method development and quantitative analysis of a minoxidil lotion 2 using FT-IR.
Materials: minoxidil PHEur (BUFA Chemicals), propylene glycol BP (Thornton & Ross), industrial methylated spirits (IMS) 74OP ketone free (HAYS), and purified water (in-house).
Analysis by FT-IR provides a rapid quantitative and qualitative method for minoxidil lotion. The calibration curve for minoxidil was linear over the concentration range studied with a regression equation y=0.286+0.890x (r=0.9998). The within day RSD of the 4 standards varied between 0.987 to 0.053. Analysis of the manufactured product provided quantitative results in the range 99.5% to 102% of theoretical. Qualitative results based on spectra fit algorithms gave full spectra results in the range 85.1 to 97.0% and measurement region fit of 100% in all cases. This method offers a very quick comprehensive analytical technique, requiring no sample preparation, that is ideal for in-process checks as well as final product analysis.
1. George WO and McIntyre PS. Infrared Spectroscopy. John Wiley & Sons 1991
back to top 
Poster 13
Hospital pharmacists suffering burnout - fact or fiction?
Surinder S Bassan
George Eliot Hospital NHS Trust, Nuneaton.
Background: "For the past two years health authorities have experienced increasing difficulties in filling posts. Again this August, at least 10 per cent of basic grade posts will remain unfilled. In some authorities the figures will exceed 20 per cent. " 2 This was reported in 1985. In 1999 not much had changed. Scott3 reported a national vacancies amounting to 13 percent. Almost three quarters of hospital pharmacies reported that unpaid additional hours worked had been greater than the previous year. Method:
Hospital pharmacy service was designed in the 1970s when activity in the evenings and week-ends was minimal. However, the change in demand was already noticeable by the mid 1980s, the Nuffield report 1 found this part-time, part week basis "unacceptable" and believed if needed pharmacy service should be available " throughout all hours of the day". The dedication and goodwill of the labour force however has been continually exploited by successive governments. Those remaining in the service have had to not only cope with ever increasing workload, but also the demoralisation of poor salaries and difficulties in recruitment and retention.
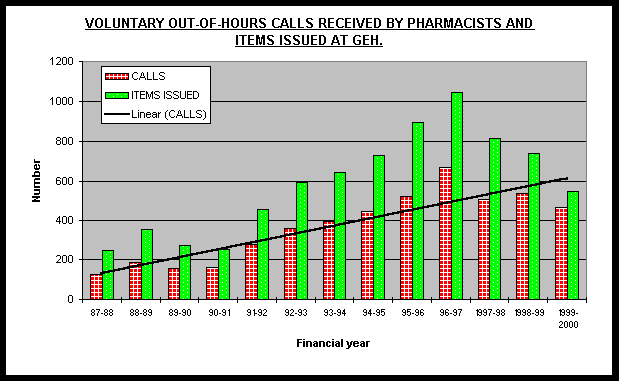
In order to assess the impact on hours worked by staff, records of hours worked were analysed retrospectively. Extra hours worked to maintain the service as well as hours worked ‘out-of-hours’ were noted. The ‘out-of-hours’ comprised of calls after 6pm during the weekdays and week-end work. The work-end hours worked included the planned opening of the Pharmacy with skeleton staff on Saturday or bank holiday mornings for which 'time-off-in lieu’ was received instead of pay. For out-of hours on call service each call was recorded. All items supplied were noted. The extra time worked for ‘time-off-in lieu’ by all staff was recorded. Some part-time staff worked additional paid hours. These have been excluded from the data reported. The number of patients discharged on a Saturday was assumed from the number of discharge (TTOs) items dispensed. Average number of items per discharged patient (worked out from a random sample) was used to work out the number of patients discharged each Saturday. Savings to the hospital in bed occupancy were calculated on the assumption that each patient with TTOs dispensed on Saturday morning resulted in two days of non-occupancy of a bed. Bed costs were assumed to be Ł150 per night.
Results:
 |
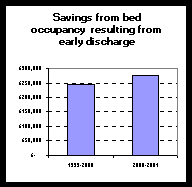 |
Conclusions: Large sums of money is saved by the hospital as a result of this voluntary unpaid work by the Pharmacy team Recommendations: References:
Data shows chronic over work by those who stay on to maintain Pharmacy Services
Just a fraction of the savings identified could be used to employ additional part-time and/or resident staff. This would provide a 7 day, all hours of the day service as envisaged by Nuffield Report and as evidence shows the NHS needs. Improved quality of care and improvements in working lives will be the immediate benefit.
1. Pharmacy: A report to the Nuffield Foundation. K Clucas, 1986, 70-71.
2. Top and bottom: Griffiths management and basic grade recruitment. "Clinicus". Pharm J, 1985, 235, 2.
3. Hospital pharmacists vacancies rate three times greater than nurses. Anon. Pharm J, 1999, 262, 99.
back to top  |
Poster 14
Managing The Introduction of New Medicines in Hospital
Dr Ray Fitzpatrick*, Dr John Mucklow**
North Staffordshire Hospital, Stoke on Trent
*Clinical Director of Pharmacy/Senior Lecturer, **Consultant Clinical Pharmacologist/ Senior Lecturer
Aims and Objectives: Methodology: The Formulary was strengthened and the pharmacy was given the mandate to apply it rigorously to all medical staff including consultants. As before, all decisions were made by the D&T committee and were based on literature evidence, considering, safety, efficacy, and cost. The use of a non-formulary drug now required the authorisation of the consultant’s clinical director. A small Medicines Management Group was formed to consider whether or not new high cost medicines could be used in the hospital. It applied the same approach as the D&T committee of independently reviewing literature evidence considering safety and efficacy, but also value for money. If the evidence supported the introduction of a new medicine, but the hospital could not afford it, the group would assist in the development of a business case for funding from the Health Authority. This was processed through the Hospital’s business planning system. Pharmaceutical advice at ward, and directorate level was also increased, and regular prescribing reports were presented at senior management meetings. Results: Following the changes, drug expenditure fell by Ł500,000 in 1998/99 and rose by only 5.6% in 1999/2000. The cost per pharmaceutical item issued in 1999/2000 was actually less than in 1993/94. Furthermore, the hospital has secured over Ł500,000 from the health authority for new medicines. Conclusion:
To develop a robust system to manage the introduction of new medicines taking into account evidence, clinical need, and cost.
The North Staffordshire Hospital has had a formulary since 1983. The content was decided by the Drug and Therapeutics (D&T) committee based on independent review of literature evidence. However, as in many hospitals the Formulary was aimed at junior staff, its objective largely educational. During 1993-1997 drug expenditure increased at an average of 6.5% (UK average 9-11%). However, with the advent of more complex HIV therapy and the hospital becoming a cancer centre, drug expenditure rose dramatically in 1997/98, and it became necessary to review the medicines management system.
The D&T committee rejected 60% of all formulary applications, approved 25% without restrictions and 15% with restrictions. The medicines management group also rejected over 60% of applications, approved 25% subject to funding and only 13% funded internally.
This work shows that a robust medicines management system can be developed and applied to all clinicians including consultants. The system can be effective in controlling the introduction of new medicines including those of high cost.
back to top  |

|